-
Posts
2,433 -
Joined
-
Last visited
-
Days Won
50
Content Type
Forums
Profiles
Gallery
Blogs
Downloads
Events
Posts posted by BusyLittleShop
-
-
4 hours ago, ShetlandBiker said:
im going to buy an endoscope so i can look in each cylinder, if it wasnt the head gasket what else should i be looking at,
Engine temps below 180ºF or 82ºC is an problem... it means that the
moisture produced during combustion is not getting hot enough to
evaporate out the pipe as steam... instead that moisture will migrate
to the oil and produce a milky white contamination...Note normal by products of combustion is water... . Every gallon of gas
creates roughly 8 pounds of water vapor... we all have witnessed water
escaping out of tail pipes on cold mornings...-
 1
1
-
-
Leaky Head Gasket Check...
You can visual inspect for a leaking head gasket by pulling the spark
plugs and peering down each hole... if the top of the piston is black
then no leak that cylinder... if the top of the piston is a bright
shiny aluminum then leak detected... because a leaky head gasket
allows coolant in and that produces steam under the heat of
combustion... the net effect is a super clean piston dome and no nasty
black...-
 2
2
-
-
I replaced my PIAA 60/55 watt Xenon gas Super White bulbs that glow in the 4200
Kelvin range with Speed Metal's 25 watt Cree LED H4 with a working high
and low beam...At the time Speedmetal LED kits were $69 each at Cycle Gear...



Out back the twin tail light bulbs are replaced with dual round circuit
boards of LED... a plug and play system sourced at WSB Laguna Seca...
RC45 LED Custom Tail Lights...Adapting the LED to center inside the existing housing requires some
mild fabrication...


Behold LED tail lights...

Hit the stop lights and now you're really shinning.. in fact the
intensity hurts the eye...

Up grading to LED and you're riding so bright you gotta were shades...-
 2
2
-
-
If you see *continuous* temps higher than 220ºF or below 180ºF then
trouble shooting is in order:Continuous engine temps above 220ºF or 104ºC is also a problem and the proper
order of items to trouble shoot are:1)Faulty radiator cap... system should hold 1.1 pressure ratio...
2)Insufficient coolant...
3)Passages blocked in the radiator, hose or water jacket...
4)Air in the system...
5)Thermostat stuck closed...
6)Faulty temp meter or thermo sensor...
7)Faulty fan...
8)Faulty fan switch...Engine temps below 180ºF or 82ºC is an problem... it means that the
moisture produced during combustion is not getting hot enough to
evaporate out the pipe as steam... instead that moisture will migrate
to the oil and produce a milky white contamination...Note normal by products of combustion is water... . Every gallon of gas
creates roughly 8 pounds of water vapor... we all have witnessed water
escaping out of tail pipes on cold mornings...The sequence of events to trouble shoot are:
1)Faulty temp meter...
2)Thermostat stuck open...
3)Faulty fan switch... (stuck on) -
I hear a moan like tire noise or a drive chain too tight... check pressure and slack...
-
 1
1
-
-
On 5/23/2025 at 9:03 PM, lawnmowerman said:
im thinking its the plates but can i .clean and reuse plates ?
Yes... plates can be cleaned and reused...
First inspect the friction plates for glazing... make sure you have plenty
of material to work with... your shop manual states clutch thickness
in thousands of an inch or mm...
Next removed the contaminants with Acetone... pick a hard surface to lay
over a 600 grit black dry emery paper... rotate the clutch plate in a
circle... you're just busting the glaze... don't get carried away
remove too much material... You should end up with a friction plate
looks dull like a new one as opposed to a shinny glazed one... recheck
thickness...
Finally check the pressure plates for bluing caused by localized heat...
make sure they are not warped... consult the manual for a thickness
range... now removed the contaminants with Acetone and wire wheeled
them to erased the blue and also to generally scuff up the surface...
you should end up with a dull surface free of Blue marks...
Steps to check your clutch for drag... your gears can't shift
smoothly if your clutch is part way engaged...
1 Place your bike on the center stand...2 Start engine and establish a warm steady idle...
3 Squeeze in the clutch lever and shift into first gear...
4 Hold in the clutch lever and note if the rear wheel coast to stop...
if it continues spinning trouble shoot the lever for travel and mastercylinder for condition... open the oil filler and look at the clutch pack
to note just how far the plates spread apart when you squeeze the lever...
Ultimately you want the rear wheel to coast to a stop when the engine
is idling and first gear selected with the clutch lever is squeezed in...Bike on its side is a simple way to shift the oil level to an angle in order to remove the clutch cover without spillage...
Have a new gasket standing by...

-
 1
1
-
-
If you see *continuous* temps higher than 220ºF or below 180ºF then
trouble shooting is in order:Continuous engine temps above 220ºF or 104ºC is a problem and the proper
order of items to trouble shoot are:1)Faulty radiator cap... system should hold 1.1 pressure ratio...
2)Insufficient coolant...
3)Passages blocked in the radiator, hose or water jacket...
4)Air in the system...
5)Thermostat stuck closed...
6)Faulty temp meter or thermo sensor...
7)Faulty fan...
8)Faulty fan switch...Engine temps below 180ºF or 82ºC is also a problem... it means that the
moisture produced during combustion is not getting hot enough to
evaporate out the pipe as steam... instead that moisture will migrate
to the oil and produce a milky white contamination...Note normal by products of combustion is water... . Every gallon of gas
creates roughly 8 pounds of water vapor... we all have witnessed water
escaping out of tail pipes on cold mornings...The sequence of events to trouble shoot are:
1)Faulty temp meter...
2)Thermostat stuck open...
3)Faulty fan switch... (stuck on)-
 1
1
-
-
Ths is the method of aluminum fork polishing I used to returned
the factory luster of my RC30 forks...1)Strip the factory clear coat with paint stripper...
2)Sand with fine foam sanding block in one direction only... you want
to curve around the tube like Honda did and not sand down the length
of the tube...3)Polish with 3M gray micro fine pad... again only polish in one
direction only... you want to curve around the tube like Honda did and
not sand down the length of the tube...4)Spray with clear coat...



-
 1
1
-
 1
1
-
-
-
-
As the RC45 Club's tech advisor I received this urgent photo and a
request as to the reason why fire almost claimed this members RC45 ...
Story goes that Bob and Pam Solloway of Coventry UK... a couple of RC
freaks... were on the way to Misano WSB... Pam's RC45 caught fire
while stopped at a light... the flames were beat back by bottle water
from a passing van... they were lucky the incident did not happen out
in the middle of nowhere for it would have spread rapidly to the fuel
lines and then the tank... nothing is worst as watching bike
flambeau...First call was to check the alternator quick disconnect at the
transformer rectifier... I was positive that the QD had suffered
enough resistance to heat and melt the insulation on the wires... the
wires got enough to start a fire which all most consumed her prize...
I seen this problem before...As I prepared my case for Pam I pulled the seat cowl off my RC45 to
send her a pic of the quick disconnect in question... Mercy was I ever
in for a shock... my quick disconnect was bad and about to turn my
bike into a crispy critter...
-
Basics of Rake...
Rake is defined as the angle of the steering head with respect to a line drawn perpendicular to the
ground (left). A smaller angle, or less rake, is sometimes referred to as being steeper. Less rake =
quicker steering and less straight line stability whereas more rake = heavier steering and more straight
line stability...Basics of Trail...
Trail gives a motorcycle stability because of the self-centering effect caused by the front wheel being
behind (or trailing) the steering axis. Too little trail, and this self-centering effect is decreased to the
point of instability. Too much trail, and the effect is so great that steering becomes heavy.Stock VFR
Rake 25.5º
Trail 95mm
Shimming the rear shock 6mm will change rake and trail towards these numbers...
Mod VFR
Rake 24.5º
Trail 92mm
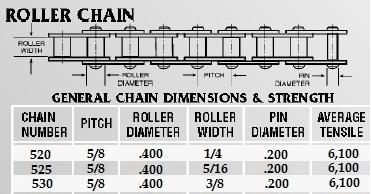
Count the number of chain links to insure you have 110... if you have `108 your cassette will cost you ride height because it is not at the 6 O clock position...
Wrong... lost ride height...

Right... correct ride height...

-
 1
1
-
 1
1
-
-
-
-
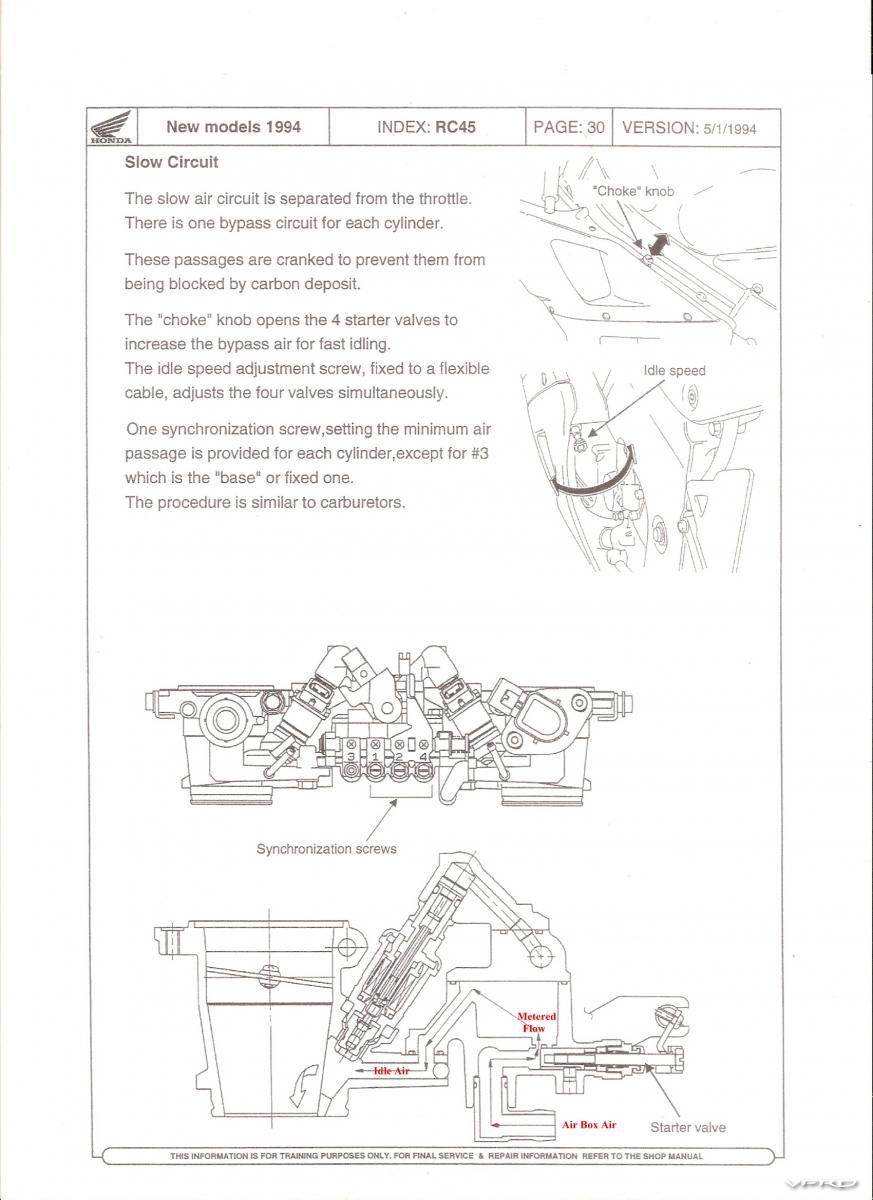
For the system to work as designed on start up the butterflies must be 100%
closed... this is because close butterflies equals sufficient vacuum... without
sufficient vacuum the fuel cannot be drawn into the combustion chamber to
support start up... the result is hard starting... the drill for cold or hot start is1 100% close throttle for sufficient vacuum
2 Full choke on when cold no choke when hot
3 Hit startEliminate the possibility that the throttle body butterflies are not
closing 100%... if any any air is bleeding pass the butterflies then
the metered flow of the idle air can not draw sufficient fuel to support combustion...
-
 1
1
-
-
On 11/24/2024 at 7:44 AM, RdChinoy said:
10. Flapper is a noise reduction device from what I can tell.
Negative...
The flapper is necessary to tune the airbox so the engine doesn't hit
any sour notes through the RPM range... Honda engineered it like a
finely tuned instrument to optimize intake efficiency and I don't
advise any owner to remove the flapper... In fact my RC45 flapper isstill installed and working to tune the airbox...
Kevin Cameron explains
What Is The Secret Importance of a Motorcycle Airbox?
If you have ever had the gas tank off your late-model sportbike, you
will notice that the front of the fuel tank doesn’t hold fuel; it
holds an airbox. In the old days, when you bought a new bike, it had
an air-filter case attached to feed the carburetors or the
fuel-injection intakes. All the sharp, young guys would immediately
rip off the filter case and replace it with four sock filters. Reduced
airflow resistance. Much better performance.One day in the late 1980s, they began to rip off the airboxes of their
new bikes and their engines fell on their faces. They lost a bunch of
performance. “This can’t be happening! Putting on sock filters always
worked before.” But it turns out the industry found a way to boost
performance by making what is known as a resonant airbox.We have all in an idle moment blown across the mouth of a beer bottle
and heard the "whoooo" of the bottle resonance. As air goes across the
mouth of the bottle, it creates a low pressure, which causes air to
flow up. That deflects the air away from the mouth of the bottle. Then
the air goes back in, the airflow from your mouth goes back across,
and the cycle repeats, rapidly fluttering and producing that deep
tone. The compressible air in the bottle is acting as a spring, and
the slug of air in the neck of the bottle is the mass that vibrates
against that spring.This intake airbox from a Honda CBR600RR is just a glorified beer
bottle. Instead of the engine blowing across the mouth of it, its four
throttle bodies are sucking from the box, pulling its pressure down.
Air rushes in through the ducts in the fairing to fill up that low
pressure. The next cylinder sucks the bottle pressure down and more
air rushes in and restores the pressure. If the volume of the box and
the mass of the air in the intake pipes are correctly chosen, the box
will hum like the beer bottle.The trick is to get your engine to draw air from the box when the
pressure is up and then the box refills when the pressure is down. And
that is why ripping the airboxes off and putting on old-time sock
filters resulted in a reduction in performance. In a specific zone of
rpm, a resonant airbox can boost your engine’s torque by 10 percent.
That’s worth having!-
 1
1
-
-
Pressurize is not the worry... vacuum is... because if the vent plugs the fuel pump
will create enough vacuum to collapse the tank... I've witnessed a VFR tank collapse
after the gas cap vent clog...

-
 1
1
-
-
Honda's V3 aided with a electric supercharger will be most noticeable in the rarefied air of high mountain passes...
-
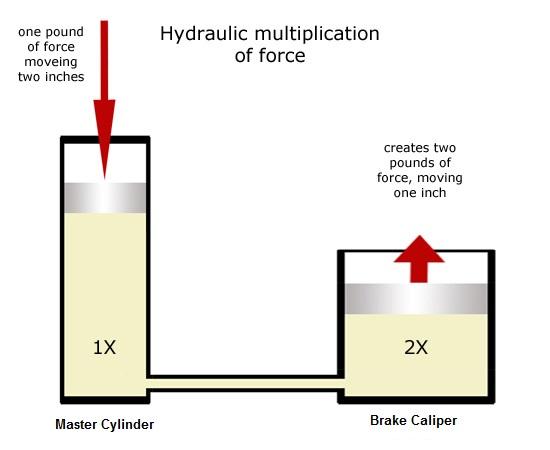
Brembo 17 or 19 numbers correspond to piston bore... the bore diameter determines how the lever feels.
Bore:
The bigger the area of the bore, the less pressure is generated
for a given input lever force. The smaller the bore area, the more
pressure can be generated from that same input force...
The bigger the area of the bore, the less stroke required to move a
given volume. The smaller the bore area, the more stroke is required
to move that same volume... -
10 hours ago, JZH said:
Well, I've always preferred the RC45 to the RC30 and I'm about to sell my house in London, so maybe...

Ciao,
JZH
Me too...
Going from an RC30 to an RC45 is like going from a Jack to King... HRC
improved everything... they transformed the analog RC30 into the digital RC45...-
 1
1
-
 1
1
-
-
Start with the battery for it's the weakest link in the whole system... To
determine the condition of an Maintenance Free battery give it a
refreshing charge... wait 30 minutes... measure terminal voltage...12.8 or higher is a good battery...
12.0 to 12.8 is a insufficient charge... recharge...
12.0 or lower... battery unserviceable... -
If you see *continuous* temps higher than 220ºF or below 180ºF then
trouble shooting is in order:Continuous engine temps above 220ºF or 104ºC is a problem and the proper
order of items to trouble shoot are:1)Faulty radiator cap... system should hold 1.1 pressure ratio...
2)Insufficient coolant...
3)Passages blocked in the radiator, hose or water jacket...
4)Air in the system...
5)Thermostat stuck closed...
6)Faulty temp meter or thermo sensor...
7)Faulty fan...
8)Faulty fan switch...Engine temps below 180ºF or 82ºC is also a problem... it means that the
moisture produced during combustion is not getting hot enough to
evaporate out the pipe as steam... instead that moisture will migrate
to the oil and produce a milky white contamination...Note normal by products of combustion is water... . Every gallon of gas
creates roughly 8 pounds of water vapor... we all have witnessed water
escaping out of tail pipes on cold mornings...The sequence of events to trouble shoot are:
1)Faulty temp meter...
2)Thermostat stuck open...
3)Faulty fan switch... (stuck on)-
 1
1
-
-
I run a 30 grade all year because it gives the right flow at the
normal engine operating temperature of 212ºF and that would be the
viscosity of 10 at operating temps... so that means for every 1000
rpms increase of oil pressure increases another 10 psi... a 30 grade
flows more oil at higher rpms which flows more oil between the
critical bearings which carries away more heat and I'm not wasting HP
just pumping oil through the blow off valve... 40 grade and 50 grade
builds pressure at the expense of flow and I wastes HP by blowing oil
through the pressure relief valve...I went to trouble to installed a digital oil pressure gauge on my RC45
to know... 30 grade at 8000 rpms 82 Psi close enough to perfect...30wt psi
1000 10
2000 20
3000 30
4000 40
5000 50
6000 60
7000 70
8000 80
9000 90
10000 99
11000 99 blow off by the pressure relief valve40wt psi
1000 12
2000 24
3000 36
4000 48
5000 72
6000 84
7000 96
8000 99 blow off by the pressure relief valve
9000 99
10000 99
11000 9950wt psi
1000 15
2000 30
3000 45
4000 60
5000 75
6000 90
7000 99 blow off by the pressure relief valve
8000 99
9000 99
10000 99
11000 99RC45 coolant 212F oil 85 psi @ 8,000 rpm

RC45 coolant 203F oil 10 psi @ 1,000rpm

-
 1
1
-
 1
1
-
-
Patrick!!! how are doing???




























1999 vfr 800 clutch sticking
in Maintenance Questions
Posted
More likely the cause of Condensation is short trips or riding in cold, when the coolant operates below 180ºF... also humid weather can cause condensation to build up inside the engine, which can then mix with the oil.